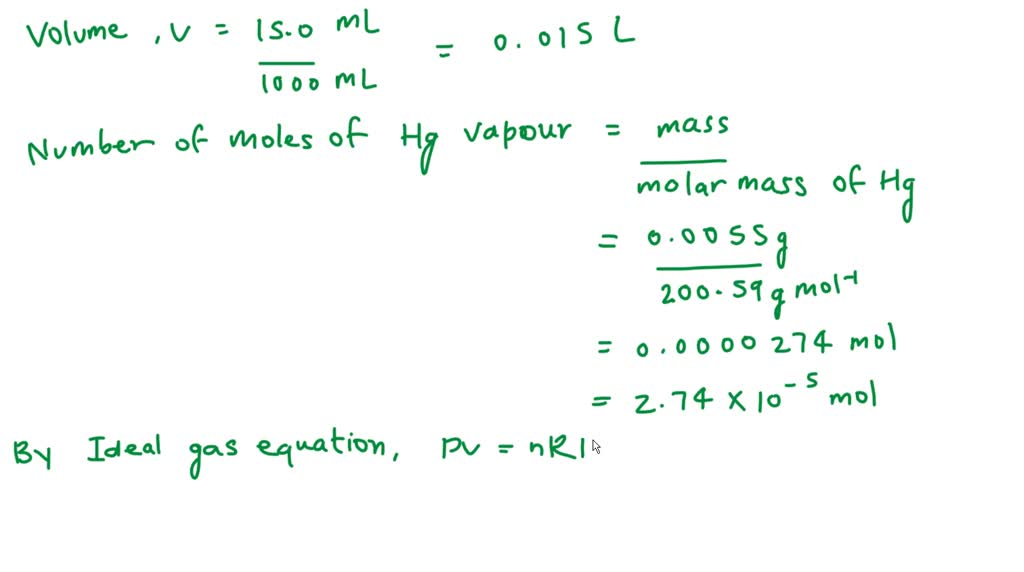
SOLVED: A mercury lamp contains 0.0055 g of Hg vapor in a volume of 15.0 mL. If the operating temperature is 2,800 K, what is the pressure of the mercury vapor?

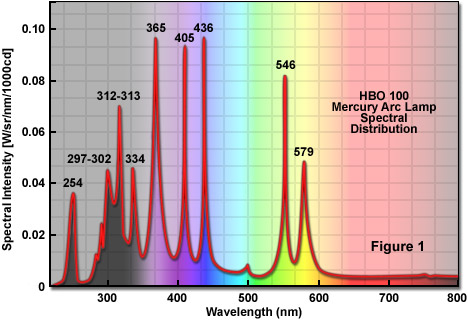
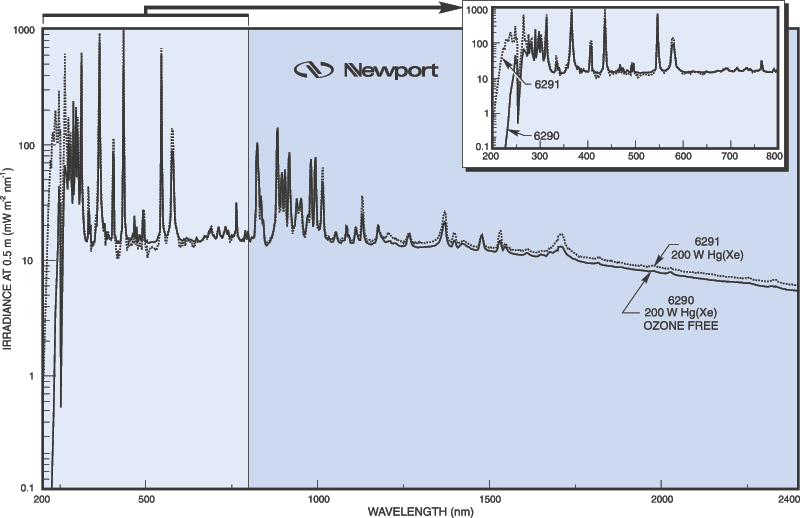
Mercury vapor lamps, low-pressure, high-pressure, optical spectrum, applications, lighting, projection, efficiency

















![Philips Cermalux 100W HG Lamp C100S54/M [Lot of 4] NOS – governmenttransitsurplus.com Philips Cermalux 100W HG Lamp C100S54/M [Lot of 4] NOS – governmenttransitsurplus.com](https://cdn.shopify.com/s/files/1/0021/6629/2533/products/img0624_78a76772-d344-4e1f-a242-d7c15cee3b31_1024x1024@2x.jpg?v=1679594723)